Abstract
Background/Methods. Marfan syndrome (MFS) is a heritable connective tissue disorder usually caused by a mutation in the fibrillin 1 (FBN1) gene. Typical characteristics of MFS that have been described include dolichostenomelia, ectopia lentis and aortic root dilatation. However, there is great clinical variability in the expression of the syndrome’s manifestations, both between and within families. Here we discuss the clinical variability of MFS by describing a large fourgeneration Dutch family with MFS.
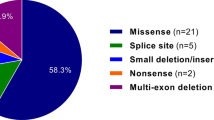
Results. Nineteen individuals of one family with a single missense FBN1 mutation (c.7916A>G) were identified. The same mutation was found in one unrelated person. Clinical variability was extensive and not all mutation carriers fulfilled the diagnostic criteria for MFS. Some patients only expressed mild skeletal abnormalities, whereas aortic root dilation was present in eight patients, an acute type A aortic dissection was recorded in two other patients, and a mitral valve prolapse was present in eight patients. In some patients cardiac features were not present on initial screening, but did however develop over time.
Conclusion. MFS is a clinically highly variable syndrome, which means a meticulous evaluation of suspected cases is crucial. Mutation carriers should be re-evaluated regularly as cardiovascular symptoms may develop over time. (Neth Heart J 2010;18:85–9.)

Similar content being viewed by others
References
De Paepe A, Devereux RB, Dietz HC, Hennekam RC, Pyeritz RE. Revised diagnostic criteria for the Marfan syndrome. Am J Med Genet. 1996;62:417–26.
Rand-Hendriksen S, Lundby R, Tjeldhorn L, Andersen K, Offstad J, Semb SO, et al. Prevalence data on all Ghent features in a cross-sectional study of 87 adults with proven Marfan syndrome. Eur J Hum Genet. 2009; ahead of print: 1–9.
De Backer J, Loeys B, Leroy B, Coucke P, Dietz H, De Paepe A. Utility of molecular analyses in the exploration of extreme intrafamilial variability in the Marfan syndrome. Clin Genet. 2007;72:188–98.
Giusti B, Porciani MC, Brunelli T, Evangelisti L, Fedi S, Gensini GF, et al. Phenotypic variability of cardiovascular manifestations in Marfan syndrome. Possible role of hyperhomocysteinemia and C677T MTHFR gene polymorphism. Eur Heart J. 2003;24:2038–45.
Hutchinson S, Furger A, Halliday D, Judge DP, Jefferson A, Dietz HC, et al. Allelic variation in normal human FBN1 expression in a family with Marfan syndrome: a potential modifier of phenotype? Hum Mol Genet. 2003;12:2269–76.
Pearson GD, Devereux R, Loeys B, Maslen C, Milewicz D Pyeritz R. Report of the National Heart, Lung, and Blood institute and National Marfan Foundation Working Group on research in Marfan syndrome and related disorders. Circulation. 2008;118:785–91.
Dietz HC, Cutting GR, Pyeritz RE, Maslen CL, Sakai YL, Corson GM, et al. Marfan syndrome caused by a recurrent de novo missense mutation in the fibrillin gene. Nature. 1991;352:337–9.
Gray JR, Bridges AB, West RR, McLeish L, Stuart AG, Dean JCS, et al. Life expectancy in British Marfan syndrome populations. Clin Genet. 1998;54:124–8.
Mizuguchi T, Collod-Beroud G, Akiyama T, Abifadel M, Harada N, Morisaki T, et al. Heterozygous TGFBR2 mutations in Marfan syndrome. Nat Genet. 2004;36:855–60.
Singh KK, Rommel K, Mishra A, Karck M, Haverich A, Schmidtke J, et al. TGFBR1 and TGFBR2 mutations in patients with features of Marfan syndrome and Loeys Dietz syndrome. Hum Mutat. 2006;27:770–7.
Sakai H, Visser R, Ikegawa S, Ito E, Numabe H, Watanabe Y, et al. Comprehensive genetic analysis of four relevant genes in 49 patients with Marfan syndrome or Marfan-related phenotypes. Am J Med Genet. 2006;140:1719–25.
Mátyás G, De Paepe A, Halliday D, Boileau C, Pals G, Steinmann B. Evaluation and application of denaturing HPLC for mutation detection in Marfan syndrome: Identification of 20 novel mutations and two novel polymorphisms in the FBN1 gene. Hum Mutat. 2002;19:443–56.
Neptune ER, Frischmeyer PA, Arking DE, Myers L, Bunton TE, Gayraud B, et al. Dysregulation of TGF-β activation contributes to pathogenesis in Marfan syndrome. Nat Genet. 2003;33:407–11.
Annes JP, Munger JS, Rifkin DB. Making sense of latent TGFbeta activation. J Cell Sci. 2003;116:217–24.
Ng CM, Cheng A, Myers LA, Martinez-Murillo F, Jie C, Bedja D, et al. TGF-beta dependent pathogenesis of mitral valve prolapse in a mouse model of Marfan syndrome. J Clin Invest. 2004;114:1586–92.
Habashi JP, Judge DP, Holm TM, Cohn RD, Loeys BL, Cooper TK, et al. Losartan, an AT1 antagonist, prevents aortic aneurysm in a mouse model of Marfan syndrome. Science. 2006;312:117–21.
Faivre L, Masurel-Paulet A, Collod-Béroud G, Callewaert BL, Child AH, Stheneur C, et al. Clinical and molecular study of 320 children with Marfan syndrome and related type I fibrillinopathies in a series of 1009 probands with pathogenic FBN1 mutations. Pediatrics. 2009;123:391–8.
Faivre L, Collod-Béroud G, Child A, Callewaert B, Loeys BL, Binquet C, et al. Contribution of molecular analyses in diagnosing Marfan syndrome and type I fibrillinopathies: an international study of 1009 probands. J Med Genet. 2008;45:384–90.
Robinson PN, Arteaga-Solis E, Baldock C, Collod-Béroud G, Booms P, De Paepe A, et al. The molecular genetics of Marfan syndrome and related disorders. J Med Genet. 2006;43:769–87.
Collod-Béroud G, Le Bourdelles S, Ades L, Ala-Koko L, Booms P, Child A, et al. Update of the UMD-FBN1 mutation database and creation of an FBN1 polymorphism database. Hum Mutat. 2003;22:198–208.
De Backer J, Loeys B, Leroy B, Coucke P, Dietz H, De Paepe A. Utility of molecular analyses in the exploration of extreme intrafamilial variability in the Marfan syndrome. Clin Genet. 2007;72:188–98.
Van Dijk FS, Hamel BC, Hilhorst-Hofstee Y, Mulder BJM, Timmermans J, Pals G, et al. Compound-heterozygous Marfan syndrome. Eur J Med Genet. 2009;52:1–5.
Author information
Authors and Affiliations
Corresponding author
Additional information
Department of Cardiology, Thorax Centre, University Medical Center Groningen, University of Groningen, Groningen, the Netherlands
Department of Genetics, University Medical Center Groningen, University of Groningen, Groningen, the Netherlands
Department of Clinical Genetics, VU University Medical Center Amsterdam, the Netherlands
Department of Human Genetics, Radboud University Nijmegen Medical Centre, the Netherlands
Department of Clinical Genetics, Leiden University Medical Center, Leiden University, the Netherlands
Department of Clinical Genetics, Academic Medical Center, University of Amsterdam, the Netherlands
Department of Cardiology, Academic Medical Center, University of Amsterdam, the Netherlands
Rights and permissions
About this article
Cite this article
Aalberts, J.J.J., Schuurman, A.G., Pals, G. et al. Recurrent and founder mutations in the Netherlands. NHJL 18, 85–89 (2010). https://doi.org/10.1007/BF03091743
Issue Date:
DOI: https://doi.org/10.1007/BF03091743